Dexketoprofen trometamol
Dexketoprofen trometamol
CAS: 156604-79-4
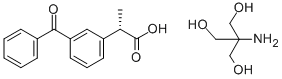
Molecular Formula: C16H14O3.C4H11NO3
Dexketoprofen trometamol - Names and Identifiers
Dexketoprofen trometamol - Physico-chemical Properties
| Molecular Formula | C16H14O3.C4H11NO3 |
| Molar Mass | 375.416 |
| Melting Point | 104.8-105.1° |
| Boling Point | 431.3℃ at 760 mmHg |
| Storage Condition | 2-8℃ |
Dexketoprofen trometamol - Clinical observation of dexketoprofen tromethamine in the treatment of herpes zoster neuralgia
from VIP Journal Professional Edition
Author:
Abstract:
objective to observe the efficacy and safety of dexketoprofen tromethamine in the treatment of herpes zoster neuralgia. Methods 132 patients with herpes zoster were randomly divided into experimental group (71 cases) and control group (61 cases). The experimental group was treated with acyclovir 0.2g,5 times a day, dexketoprofen tromethamine 25mg, the control group was treated with acyclovir 0.2g,5 times/d and ibuprofen 0.3g,3 times/d, respectively. The curative effect and adverse reactions were observed on the 3rd, 5th and 7th day of treatment. Results dexketoprofen tromethamine combined with acyclovir can significantly improve the pain symptoms of the patients. There was no significant difference in the effective rate between the two groups (P>0.05). The incidence of adverse reactions was 7.46% in the experimental group and 20.33% in the control group, the difference was significant (P<0.05). Conclusion dexketoprofen tromethamine combined with acyclovir treatment. Can effectively relieve herpes zoster neuralgia, and adverse reaction rate is low.
Key words:
dexketoprofen tromethamine; Ibuprofen; Herpes zoster
DOI:
CNKI:SUN:ZBFX.0.2009-06-014
cited:
year:
2009
Dexketoprofen trometamol - Study on quality standard of dexketoprofen tromethamine sustained-release double-layer tablets
from Palm Bridge research
Author:
Li yuetin , Hou Jingyu , Hu Jie , Pan Jie , Huang Yong , Zheng Lin
Abstract:
Objective: to establish the quality standard of dexketoprofen tromethamine (DKPF) sustained release tablets. Methods: The content of DKPF in 3 batches of DKPF sustained-release tablets was determined by UV spectrophotometry, and, the release rate and stability in different media were evaluated. Results: the established UV method for the determination of D-ketoprofen in DKPF sustained-release tablets had a good linear relationship with the absorbance in the range of 1.19~21.49 mg/L, A = 0.042 3 C 0.002,r = 0.999 9, the recovery rate was 99.8% ~ 100.6%,RSD was 0.34% ~ 1.02%; The release behavior of double layer tablets was in accordance with the first order kinetic equation; the results of dissolution test showed that the 3 batches of samples could release the drug rapidly within 30 min, and then release slowly, reaching the requirement of rapid release and sustained release, thermal stability, moisture absorption is serious under the condition of humidity 75%. Conclusion: The UV method can effectively monitor the quality of DKPF sustained-release bilayer tablets. The method is simple and reliable.stowed
Key words:
dexketoprofen tromethamine UV spectrophotometry Quality Standard dissolution release
year:
2015
Dexketoprofen trometamol - Pharmacokinetics of dexketoprofen tromethamine thermosensitive hydrogel in rats
from Palm Bridge research
Author:
Zheng Lin , plum lobe , Lu Yuan , Li yuetin , benzupeng , Chen Siying , Huang Yong
Abstract:
Objective: to establish a UPLC-MS/MS Method for the pharmacokinetic study of dexketoprofen tromethamine thermosensitive hydrogel in rats. Methods: A Waters BEH C18(50mm × 2.1, 1.7 μm) column was used with the mobile phase of 0.1% formic acid acetonitrile-0.1% formic acid aqueous solution by gradient elution; electrospray ionization source (ESI) was used for mass spectrometry, and scanning mode was multi-reactive ion monitoring (MRM). The blood concentration of dexketoprofen tromethamine was determined after subcutaneous injection of hydrogel solution, subcutaneous injection of drug solution (0.9% NaCl solution) and intragastric administration (ig) in rats, the related pharmacokinetic parameters were calculated. Results: compared with the injection solution, the Cmax of the injected hydrogel was significantly decreased, and the Tmax was more prolonged. Compared with the gavage (ig), the AUCo-∞ of the injected hydrogel was significantly increased, and the Cmax was decreased, tmax was prolonged and half-life was significantly increased. Conclusion: The method is specific, rapid, accurate and sensitive, and can be used for pharmacokinetic study of D-ketoprofen tromethamine thermosensitive hydrogel in rats.stowed
Key words:
dexketoprofen tromethamine thermosensitive hydrogel pharmacokinetics LC-MS/MS
year:
2016
Supplier List
CAS: 156604-79-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 156604-79-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 156604-79-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 156604-79-4
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 156604-79-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 156604-79-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 156604-79-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 156604-79-4
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 156604-79-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 156604-79-4
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 156604-79-4
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 156604-79-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025





